publications
Synthesis, Photophysical, and Chiroptical Properties of Optically Active BINOL-Boranil Enantiomers
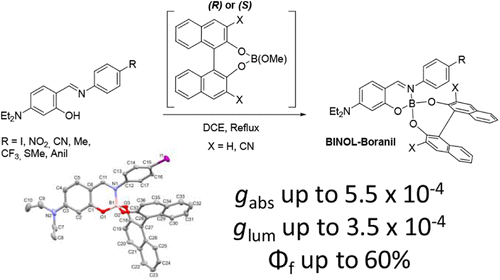
We describe a very simple and efficient gram-scale synthetic pathway toward Boranils substituted directly on the boron center by 1,1′-bi-2-naphthol derivatives (BINOL). Properties of the resulting BINOL-Boranils were investigated by nuclear magnetic resonance (NMR), UV–visible, fluorescence, electronic circular dichroism (ECD), and circularly polarized luminescence (CPL) spectroscopies. Insight into the molecular structure is also provided based on single-crystal X-ray diffraction experiments. These chiral dyes typically feature high molar absorption coefficients in the range of 35,000 to 90,000 M–1·cm–1, absorption dissymmetry factors (gabs) in the range of 2.5–5.5 × 10–4 at the maximum wavelength of absorption of Boranils, and CPL brightness up to BCPL = 5.4 M–1·cm–1.
Samuel Decorps, Rayan Hachem, Juichi Bainvel-Sato, Rémy Devienne, Bruno Baguenard, Stéphan Guy, Erwann Jeanneau, Floris Chevallier, Christophe Bucher, Laure Guy,* Denis Frath* ACS Omega 2026, 11, 21816–21823, 10.1021/acsomega.5c11819
Comments are closed